 Currently the number one non-surgical aesthetic treatment in the U.S., Botox® is also the leading brand of botulinum toxin type - A in terms of documented clinical trials and practical use around the world.
Currently the number one non-surgical aesthetic treatment in the U.S., Botox® is also the leading brand of botulinum toxin type - A in terms of documented clinical trials and practical use around the world.
Botox®, manufactured by Allergan, was the first botulinum toxin to gain a cosmetic licence endorsing its use for glabellar lines and wrinkles in the US in 2002. It was also eventually licenced for this use in the U.K. in March 2006 under the new brand name of Vistabel® (with a vial dose specific to treat glabellar lines) and was the first brand of botulinum toxin to receive a license for cosmetic use in this country.
Although Botox® is still readily available and used by many UK cosmetic clinics, Vistabel® (although identical in formulation to Botox) is technically the first brand of botulinum toxin type A licensed for cosmetic use in the UK.
Clostridium botulinum type - A neurotoxin complex.
Vistabel® is made from purified type - A neurotoxin produced by the bacteria Clostridium botulinum.
It temporarily prevents the release of acetycholine from sympathetic nerve terminals. To learn in detail how Vistabel® injections work, please click on Botulinum Toxin Treatment FAQ.
Prescription only medicine
It should be used by trained members of the medical profession only.
In March 2006, Vistabel® was granted a licence in the UK from the Medicines and Healthcare products Regulatory Agency (MHRA) for the "temporary improvement in the appearance of moderate to severe glabellar lines (vertical `frown` lines between the eyebrows) in adult women and men aged 65 and younger, when the severity of these lines has a psychological impact for the patient" .
Individuals with a known hypersensitivity to any component of the formulation.
Patients who have generalised disorders of muscle activity (e.g. myasthenia gravis).
Patients where aminoglycoside antibiotics or spectinomycin are already being used or are likely to be used.
Patients who have bleeding disorders of any type.
Pregnant or lactating women.
Bruising or bleeding at the site of injection, headache, and rarely eyelid ptosis or diplopia, and brow droop.
Click on Botulinum Toxin Side Effects to learn more about the risks or side effects associated with Vistabel®.
Most clinics and practitioners charge by area of the face treated, such as the glabellar lines (frown lines between the eyes).
Practitioners may charge from around £175 - £300 per treatment.
Most of the literature on Botulinum Toxin, and hence the clinical evidence and statistics, are based on Botox® as this brand represents around 80% of the world’s use of Botulinum Toxin.
Because it is a prescription only medicine; it had to undergo strict clinical trials in the U.S., where it is approved for cosmetic use, in order to gain a license. Remember, the drug has been licensed in the U.S. since the late 1980s.
The graph below shows the results of a study published in 2001.
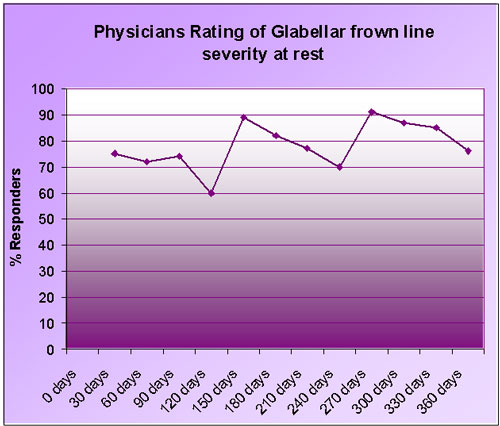
Data taken from Carruthers, Lowe, & Menter; One year study on the safety and efficacy of Botulinum toxin in Patients with Glabellar Lines; Presented at the AAD March 2001.
On the basis of this evidence, most practitioners would now recommend an initial course of Botox® treatment every 3 or 4 months for the first year. It now appears that after this, the time between future injections may lengthen, with practitioners reporting that some patients may only need to return twice a year to maintain results.
Vistabel ® is a registered trademarks of Allergan Inc.
Do you have a question? Ask one of our experts NOW