
27
Sep
2012
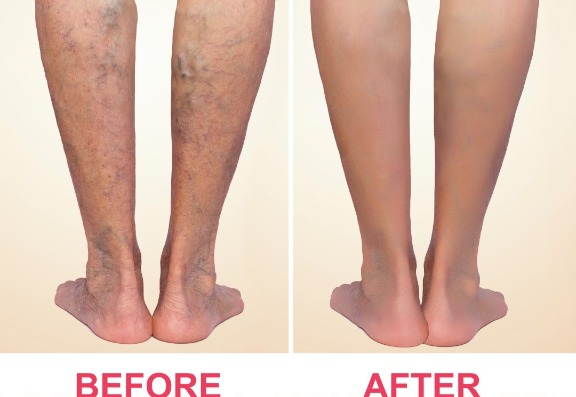
Venaseal™ or “superglue” treatment by Sapheon is a unique non-surgical treatment that uses a medical adhesive (cyanoacrylate) to safely and effectively treat varicose veins.
The technique has been developed over the last 5 years by an American company called Sapheon and last year it was granted a CE mark and it is available in Europe.
Unlike other treatments, VenaSeal™ does not require tumescent local anaesthetic around the vein, so there are far fewer injections than with treatments such as VNUS Closure or Endovenous Laser.
The VenaSeal™ procedure also eliminates the use of heat (or thermal energy) so there is virtually no risk of nerve or skin injury.
As the medical “superglue” sets within a few minutes, there is not the prolonged healing process associated with ultrasound-guided foam sclerotherapy and medical or compression stockings are not required afterwards.
The procedure is very quick (around 15 minutes).
How does the VenaSeal™ system work?
A tiny catheter is positioned inside the unhealthy vein using ultrasound through a small puncture in the skin.
Next, a small amount of adhesive is delivered inside the vein using a special dispenser to seal the vein which has been identified as being unhealthy and which is causing the varicose veins.
After the treatment, blood then travels back to the heart in nearby healthy veins.
The glued vein is then absorbed by the body’s natural healing processes.

Is the VenaSeal™ Adhesive used during the procedure safe?
The VenaSeal™ Adhesive that is used during the procedure is made with a medically approved type of Cyanoacrylate, which has a proven track record of over 50 years of use in the field of medicine.
The VenaSeal™ system uses a unique formulation of the compound and it is specifically designed for the treatment of varicose veins.
The adhesive is very viscous and it bonds within a few minutes so there is no risk of the adhesive travelling around the body.
Many thanks to the author of this blog Dr Haroun Gajraj who has over 30 years’ experience of treating people with varicose veins, thread veins and other vein complications such as phlebitis, varicose eczema and varicose ulcers.
He is the only doctor based in Dorset who is in a full-time private practice treating people with vein problems.
My wife and I founded The VeinCare Centre for the treatment of venous disease and we have gathered together an award-winning team of experts.

If you want to read more, the experts at Consulting Room really know what they're talking about and have put together some Varicose veins, EVLA laser treatment and sclerotherapyFAQs just for you.
If you have more questions, you can use the Varicose veins, EVLA laser treatment and sclereotherapy questions feature to talk to our panel of trained medical experts.
If you're keen to get started with any of these treatments right away then you're in luck - those clever folks also have a list of trusted, accredited Varicose veins, EVLA laser treatment, and sclerotherapy clinics in your area.
This is everything you need to know about choosing the right Microsclerotherapy Sclerosant in 2024...
Did you know that the UK is the only country in the world with an unregulated aesthetic industry?
The recent documentary by Olivia Attwood shows how confusing it can be to choose who and where to get your aesthetic treatments from, and how safe they are...
Hey, wait!
Before you go.....
Let's stay in touch, pop your details here and we'll send our editor's hand-picked updates on your fave subjects.